Basic Chemical Reactions of Polyurethane Foam
Polyurethane is sometimes referred to as PU, which is the abbreviation of polyurethane. As the name suggests, it is named after the urethane formed by the reaction of isocyanate and hydroxyl compound as its characteristic chain link. But in fact, there are many chemical reactions involved in polyurethane, especially polyurethane foam, and there are not many main reactions that are really influential. Most of the chemical reactions of polyurethane are related to the chemical properties of isocyanate NCO in isocyanates. NCO can not only react with hydroxyl compounds to form carbamates, but also react with other "active hydrogen" compounds to form different chemical links. Thereby changing the chemical bond structure and material properties of polyurethane.

The active group of isocyanate is isocyanate NCO. The electronic structure of NCO shows that it has a strong resonance effect. The usual reaction is mainly the addition reaction of carbon-nitrogen double bond. Compounds with active hydrogen first attack the nitrogen atom of NCO, and other atoms connected to the active hydrogen are added to the carbon atom of the isocyanate carbonyl group. The active hydrogen compound refers to a compound that can replace a hydrogen atom by metallic sodium, mainly including hydroxyl-containing alcohols, amino-containing amines, water, and the like.
The main reactions of polyurethane can be divided into polymerization reaction, foaming reaction and cross-linking reaction according to their functions.
1. Polymerization
That is (1) the reaction of isocyanate and hydroxyl
The NCO of isocyanate reacts with the hydroxyl OH of alcohol (usually polyether, polyester or other polyol) to form polyurethane.

2. Foaming reaction
the reaction of isocyanate and water
The NCO of isocyanate reacts with water to form unstable carbamic acid first, which is then decomposed into amine and carbon dioxide.

3. Cross-linking reaction
Including (3) allophanate reaction and (4) biuret reaction
The hydrogen on the nitrogen atom of the urethane group reacts with the NCO of the isocyanate to form an allophanate. The hydrogen on the nitrogen atom of the urea group in the diurea reacts with the isocyanate group of the isocyanate to form a biuret.

The above two reactions (3) and (4) are both cross-linking reactions. Generally speaking, the reaction rate is relatively slow. In the absence of a catalyst, the reaction should be carried out at 110-130 °C. The higher the temperature, the faster the reaction rate. In addition, because the allophanate and biuret linkers are not very stable, that is to say (3) and (4) are reversible reactions.
To sum up, there are three types of basic reactions of PU: reaction (1) is a chain extension reaction or polymerization reaction, reaction (2) is a gas generation reaction or foaming reaction, and reactions (3) and (4) are cross-linking reactions.
In the foaming process of PU, these reactions are carried out simultaneously at a relatively high speed, and most of the reactions can be completed within a few minutes under catalyst conditions. Finally, a polyurethane foam of high molecular weight and a certain degree of crosslinking is formed.
The polymerization reaction and the crosslinking reaction are the main reactions in the formation of the polyurethane foam skeleton, which can be collectively referred to as the gel reaction; while the foaming reaction is the main reaction for the increase of the volume of the polyurethane and the formation of the gas source of the hollow foam structure.
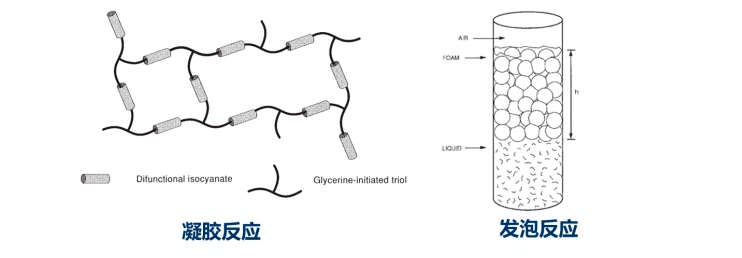
The development and adjustment of all formulations of polyurethane foam, including many practical problems in foam production, are inseparable from the balance of gel reaction and foaming reaction.












